Isotopic Molecular Imaging Group | Wilson Lab
The goal of research in the Isotopic Molecular Imaging Group (Wilson lab) is to develop metabolism and microenvironment-targeted imaging methods, to guide intervention and predict response to molecular therapies. We are currently developing cutting-edge positron emission tomography (PET) and magnetic resonance imaging (MRI) techniques to study basic biochemical pathways and their alterations in infection, cancer and other disorders. In particular, the group pursues imaging targets relevant to the central nervous system and the practice of clinical neuroradiology.
New chemistries are used in the design and synthesis of imaging probes, which have strong potential for translation into the clinic. Several tracers developed in the lab are currently being investigated in UCSF patients. We are particularly focused on the molecular imaging of infection using bacteria-specific metabolic radiotracers and MR-compatible methods employing stable isotopes.
Research
The redox status of cancer cells is critical in understanding both tumor aggressiveness and response to therapy.
The Wilson laboratory is developing hyperpolarized 13C probes that are critical in advancing and understanding dynamic nuclear polarization- nuclear magnetic resonance (DNP-NMR)…
The Wilson laboratory is developing metabolism-targeted methods to identify tumors harboring IDHm, either (1) based on consumption of tumor-relevant metabolites or (2) direct detection…
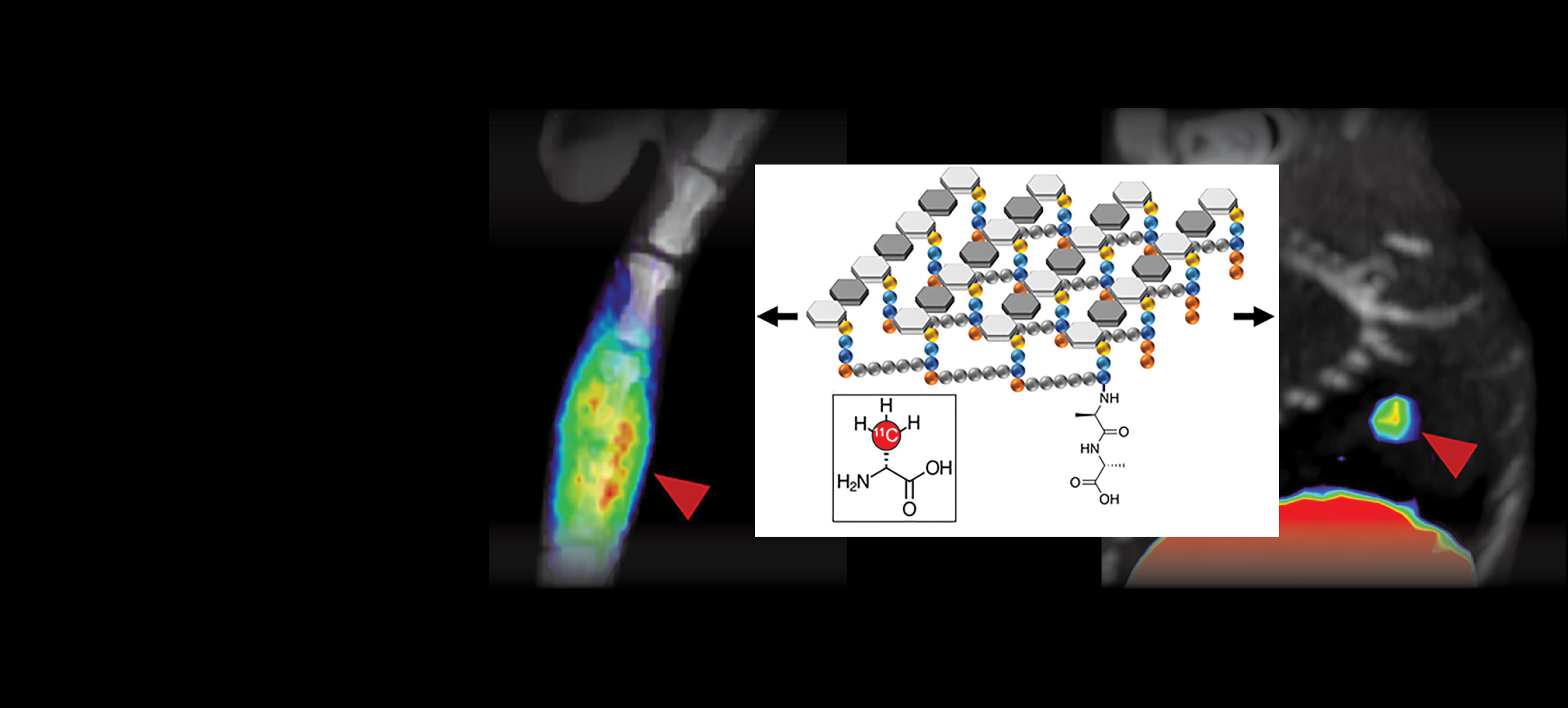
The laboratory is currently developing new ways to incorporate 11C and 18F radionuclei, expanding the arsenal of radiopharmaceuticals applied to biologic problems.
The Wilson laboratory is pursuing clinically translatable biomarkers for infection, particularly discitis-osteomyelitis, which may be difficult to diagnose using computed tomography …
We are investigating new deuterium-enriched probes for the molecular imaging of infection and Alzheimer’s disease (with Drs. Chaumeil, Gordon).
Publications
Opportunities
Awards
- R01 “First-in-human evaluation of an astrocytic glutamate transporter (EAAT2) PET tracer in healthy and Alzheimer’s diseased brain" (PIs Wilson, VanBrocklin).
- UCSF resource allocation program (RAP) grant for 18F infection imaging translational expenses.
- R01 “Imaging cerebral metabolic impairment in AD using deuterium MRI” (PIs Chaumeil, Gordon, Wilson).
- R21 “Imaging bacterial infection using deuterium-enriched sugar alcohols" (PI Wilson).
- R01 “Sensing living P. Aeruginosa using D-alanine derived radiotracers” (PIs Wilson, Ohliger, Engel).
- R01 “New radiotracer development to study immune cell mobilization of granzyme proteolytic activity” (PIs Evans, Wilson, Craik).
- R21 “Diagnosis of low-grade glial tumors using 11C glutamine isotopomers” (PIs Villanueva-Meyer, Wilson).
- R01 “ACE2-targeted PET radiotracers for investigating the spatiotemporal distribution of SARS-CoV-2 organ injury and therapy response” (PIs Wilson, Flavell, Desai).
- Cystic Fibrosis Foundation (CFF) “Mechanisms of P. Aeruginosa colonization in cystic fibrosis” (PIs Wilson, Engel).
News
- Congratulations to Dr. Matthew Parker who received a faculty position at Stony Brook University.
- Congratulations to Dr. Jaehoon Shin who is starting a faculty position in the Department of Radiology and Biomedical Imaging July 2022.
- Congratulations to Aryn Alanizi, UCSF Master of Science in Biomedical Imaging (MSBI) graduate who will be starting medical school at Eastern Virginia Medical School in summer 2022.
- Congratulations to Dr. Chao Wang who accepted a patent law position in San Diego.
- Congratulations to Dr. Hecong Qin who will be starting medical school at the University of Chicago summer 2022.
- Congratulations to Dr. Ilona Polvoy who will be continuing her internal medicine residency in Israel.
- Welcome Sanghee Lee (2022) who is a chemist/radiochemist to received his Ph.D. from Seoul National University.
- Welcome Marina Alvarez Lopez (2022) who is a microbiologist who received her Ph.D. form the University of Groningen.
- Welcome Ryan Nillo (2022) who is a staff research associate having worked previously with Drs. Sugrue and Desikan at UCSF.
- Welcome Priamo Pichardo Gonzalez (2022) who is a medical student from Puerto Rico and future diagnostic radiology resident.
- Welcome Sarah Rabbitt (2021) who is a synthetic chemist and recent graduate of the University of San Francisco.
- Dr. Polvoy’s manuscript, “Imaging joint infections using D-methyl-11C methionine PET/MRI: initial experience in humans” published in EJNMMI (2022). https://pubmed.ncbi.nlm.nih.gov/35732972/
- Dr. Polvoy’s manuscript, “Nuclear imaging of bacterial infection: state of the art and future directions” published in JNM (2020). https://jnm.snmjournals.org/content/61/12/1708
- Dr. Parker’s manuscript, “Cyclic 68Ga- labeled peptides for specific detection of human angiotensin-converting enzyme 2” published in JNM (2021). https://pubmed.ncbi.nlm.nih.gov/33637588/
- Dr. Jaehoon Shin’s manuscript, “Antigen-dependent inducible T cell reporter system for PET imaging of breast cancer and glioblastoma” accepted at JNM; preprint @ bioRxiv (2022). https://www.biorxiv.org/content/10.1101/2022.02.16.480729v1
- Dr. Jianbo Liu’s manuscript, “Synthesis of N-trifluoromethyl amides from carboxylic acids” published at Chem (2021). https://www.cell.com/chem/pdf/S2451-9294(21)00361-2.pdf
- Dr. Zhao’s manuscript, “In vivo measurement of granzyme proteolysis from activated immune cells with PET” published in ACS Central Science (2021). Collaboration with Evans, Craik labs. https://pubs.acs.org/doi/full/10.1021/acscentsci.1c00529
- Congratulations to Drs. Shin, Polvoy, Qin, Sorlin, Parker, and Alanizi for scientific abstracts accepted at WMIC, Pacifichem, ISRS, and EMIM.
Resources
Lab Members
- Aryn Alanizi, MS is a medical student at East Virginia Medical School.
- Jacob Brown, MD, PhD is a neuroradiologist at Utah Radiology Associates.
- Valerie Carroll, PhD is a Staff Scientist at EAG Laboratories.
- Rebecca Dumont-Walter, MD is a Neuroradiologist at Geneva, Canton of Geneva (Switzerland).
- Robert Flavell, MD, PhD is a UCSF Radiology and Biomedical Imaging faculty member and close collaborator.
- Tanushree Ganguly, PhD is a Project Scientist at the UCD Department of Biomedical Engineering.
- Mausam Kalita, PhD is a Specialist in the UCSF Department of Radiology and Biomedical Imaging.
- Kayvan Keshari, PhD is a MSKCC faculty member.
- Dongdong Liang, PhD is a chemistry department faculty member in China.
- Jianbo Liu, PhD is a faculty member at Sun Yat-Sen University in China.
- Wei Liu, PhD is a faculty member at the University of Cincinnati.
- Justin Luu is a PhD candidate at Emory University.
- Christopher Mutch, MD, PhD is a practicing neuroradiologist at Bay Imaging Consultants.
- Kiel Neumann, PhD is a faculty member at the University of Virginia Department of Radiology and Medical Imaging.
- Matthew Parker, PhD is an Assistant Professor at Stony Brook University.
- Ilona Polvoy, MD is an internal medicine resident at Rambam Health Care.
- Hecong Qin, PhD is a medical student at University of Chicago.
- Oren Rosenberg, MD, PhD is head of bacterial infections R&D at Johnson and Johnson Pharmaceuticals.
- Victor Sai, MD is a UCLA Diagnostic Radiology faculty member.
- Megan Stewart, PhD is a Senior Manager at Clovis Oncology.
- Celine Taglang, PhD is a Postdoctoral Scholar at UCSF.
- Andrew Taylor, MD, PhD is a UCSF Radiology and Biomedical Imaging faculty member.
- Chao Wang, PhD is a scientific advisor at Jones Day Law Firm in San Diego.
- J.P. Yu, MD, PhD is a University of Wisconsin Department of Radiology faculty member.
Contact Us
Isotopic Molecular Imaging Group (Wilson Lab)
185 Berry Street
China Basin Landing
San Francisco, CA 94107
Ph: (415) 353-1668
Fax: (415) 353-8593












