Program for Molecular Imaging & Targeted Therapy | Hope Lab
The Program for Molecular Imaging and Targeted Therapy Lab involves a number of clinical trials focused on the translation of novel targeting imaging and therapy agents. We have a strong translation clinical research focus on novel radiotracers and targeted radionuclide treatments. We also work on clinical applications of simultinaneous PET/MRI.
Research
Over the past five years I have worked on a number of projects related to neuroendocrine tumors.
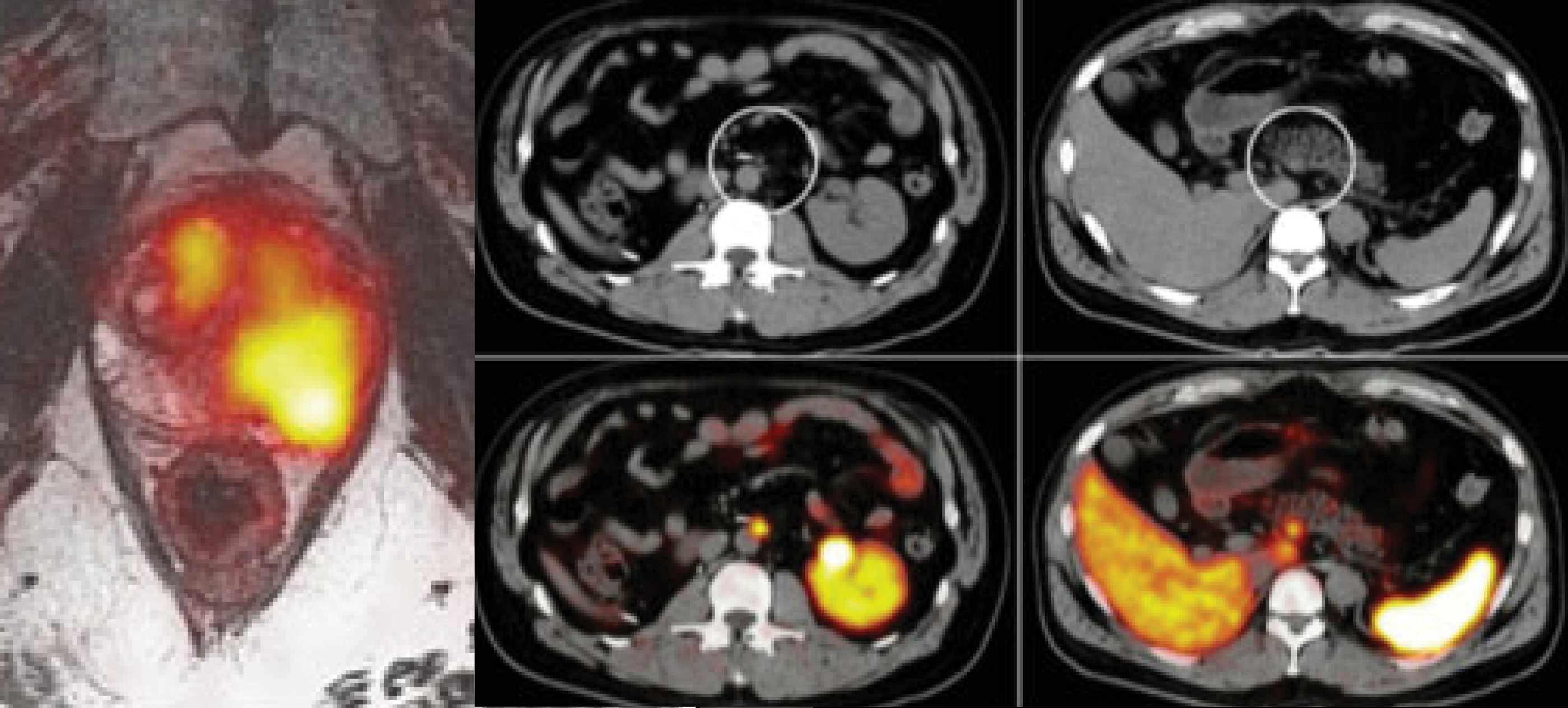
In the last few years there has been significant improvement in the imaging of prostate cancer using Prostate Specific Membrane Antigen (PSMA) PET.
The simultaneous modality, PET/MRI, is a challenging environment due to the limitations of bringing PET and MRI together, but when appropriately leveraged, PET/MRI offers significant…
Opportunities
Postdoctoral Position in Simultaneous PET/MRI
A postdoctoral appointment is available under the supervision of Dr. Peder Larson and Dr. Thomas Hope at the University of California, San Francisco (UCSF) Department of Radiology and Biomedical Imaging. This position is focused on the development of tools to evaluate the quantitative accuracy of PET/MRI. Work will involve PET reconstruction efforts, phantom development and work on novel MRI sequences to provide improved attenuation correction. Position is fully funded for five years.

