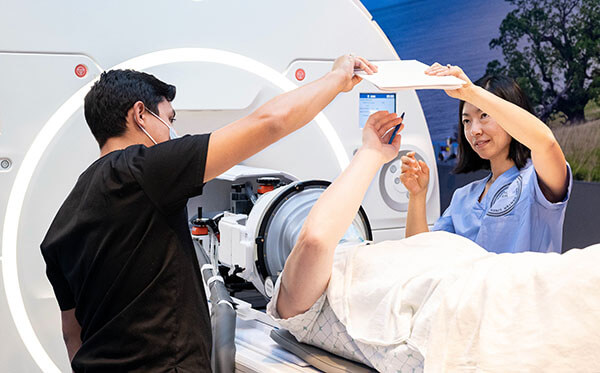
Focused ultrasound is an innovative, noninvasive treatment used clinically at UCSF to treat brain disorders such as essential tremor, bone and soft tissue tumors, and prostate cancer. Much as a magnifying glass can focus sunbeams to burn a hole in a piece of paper, our focused ultrasound technologies focus sound waves onto a tiny area, generating enough heat to destroy abnormal tissue. Our pioneering focused ultrasound program uses advanced imaging to guide these treatments with millimeter precision and is working to develop the next generation of focused ultrasound treatments for brain tumors and a range of neurologic and psychiatric diseases.