Imaging Cortical Bone Vasculature

A decade ago, Thomas Link, MD, PhD, was senior author on a paper in J Bone Miner Res1 that suggested that severe deficits in cortical bone quality are responsible for fragility fractures in postmenopausal diabetic women. Building on that work, our Bone Quality Research Lab has developed a technique to visualize intra-cortical vessels and assess the structural changes that can degrade bone strength.
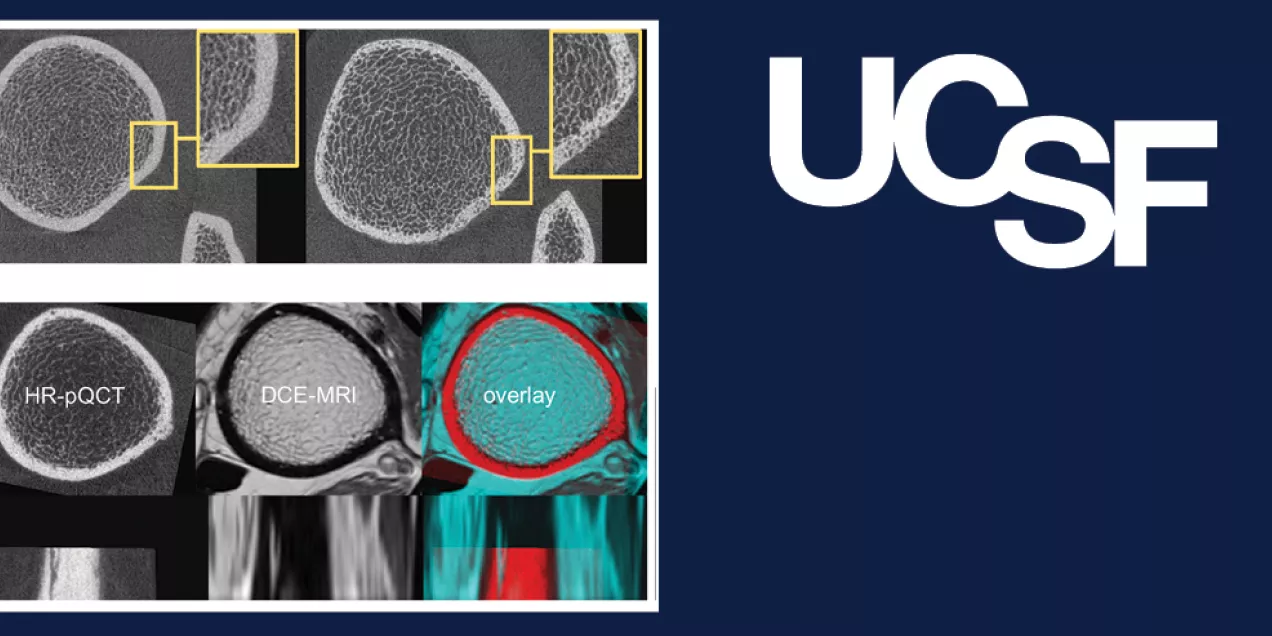
To understand how the normal vascular structure is altered, rendering the compact cortical shell very porous in some patients, our lab developed a technique that uses both dynamic contrast-enhanced MRI and high-resolution peripheral quantitative computed tomography (HR-pQCT)2. Using this new capability, we have analyzed baseline data of the distribution and size of intracortical vessels in patients with diabetes compared to healthy controls.
One imaging challenge is deducing which cortical bone pores house blood vessels and which canals are filled with fat. Using this technique, contrast is injected into the patient and multiple MRI scans are taken to track how the contrast moves through the veins, allowing researchers to image their structure.
The cortical bone microstructure itself does not show up well on MRI, but HR-pQCT can visualize the very fine details of the internal bone structure, allowing researchers to see the pores and canals in the bones through which the blood vessels could travel, while the MRI identifies which actually have vessels threaded through them. Earlier work by the Bone Quality Research Lab has documented that pores that do not contain vessels are filled with fat3.
Results indicate that patients with diabetes have fewer and larger vessels through their bone, whereas in the controls the vessels are more homogeneously distributed and small. We expect that determining the content and spatial distribution of cortical pore space will reveal the biological systems influencing pore expansion.
The overall goal of this study is to understand the longitudinal evolution of human diabetic bone disease and to investigate and visualize the underlying biological processes that drive increased cortical porosity in the setting of T2D. Filling this knowledge gap will elucidate appropriate cellular targets for drug development to prevent or reverse pathological pore development and the associated skeletal fragility.

1) Patsch JM, Burghardt AJ, Yap SP, Baum T, Schwartz AV, Joseph GB, Link TM. Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J Bone Miner Res. 2013 Feb; 28(2):313-24. doi: 10.1002/jbmr.1763
2) Löffler MT, Wu PH, Kazakia GJ. MR-based techniques for intracortical vessel visualization and characterization: understanding the impact of microvascular disease on skeletal health. Curr Opin Endocrinol Diabetes Obes. 2023 Aug 1; 30(4):192-199. doi: 10.1097/MED.0000000000000819. Epub 2023 Jun 19
Wu PH, Gibbons M, Foreman SC, Carballido-Gamio J, Han M, Krug R, Liu J, Link TM, Kazakia GJ. Cortical bone vessel identification and quantification on contrast-enhanced MR images. Quant Imaging Med Surg. 2019 Jun; 9(6):928-941. doi: 10.21037/qims.2019.05.23. PMID: 31367547; PMCID: PMC6629562
3) Garita B, Maligro J, Sadoughi S, Wu PH, Liebenberg E, Horvai A, Link TM, Kazakia GJ. Microstructural abnormalities are evident by histology but not HR-pQCT at the periosteal cortex of the human tibia under CVD and T2D conditions. Med Nov Technol Devices. 2021 Jun; 10:100062. doi: 10.1016/j.medntd.2021.100062. Epub 2021 Feb 3. PMID: 37383338; PMCID: PMC10306320