The Transition from Wake to Sleep: Watching Cortical Synchronicity and Information Flow
For most of us, falling asleep does not feel as simple as flipping a switch. The surprisingly complex process of drifting from wakefulness to sleep is governed by the brain’s cortex, and new collaborative research from UCSF imaging scientists and neurologists advances our understanding of this important phenomenon. In “Cortical Synchrony and Information Flow during Transition from Wakefulness to Light Non-Rapid Eye Movement Sleep” published in The Journal Of Neuroscience, the researchers discovered that during this period, information flow activity shifts from the back of the brain to the front, accompanied by an increase in cortical excitation relative to inhibition.
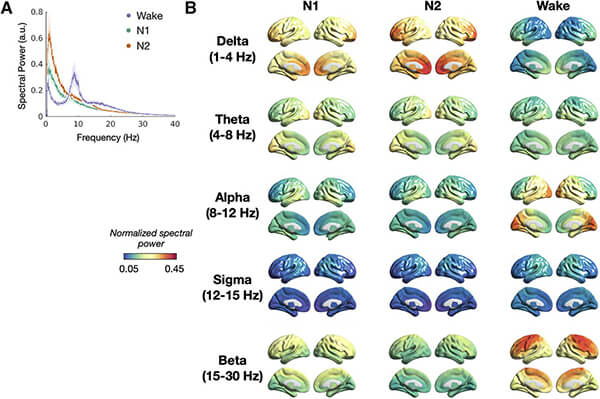
This study sheds light on the orchestration of local and long-range cortical neural structure that is fundamental to sleep onset and supports an emerging view of cortically driven regulation of sleep homeostasis. Understanding how the brain coordinates neural activity with sleep onset provides insights into both the physiological functions subserved by sleep and pathology associated with sleep onset. Previous fMRI/PET based studies contended with low temporal resolution and limited insights into neural activity, while MEG/EEG studies lack comprehensive assessments of simultaneous local and long-range synchrony and information flow during sleep onset. By using magnetoencephalography (MEG) imaging and inferring the underlying mechanisms through biophysical models, this study clarified the relationship between neural synchrony and excitation-to-inhibition balance.
As sleepiness sets in, the cortex’s regulation of the brain shifts tempo. Certain areas, like the mesial frontal region, amplify their internal rhythms, pulsating with low-frequency delta waves. These synchronized oscillations propagate outward, drawing other areas into the lower energy patterns of sleep. Long-range connections also take part, quickening the process beyond brain regions influencing their immediate “neighbors”. Bilateral frontoparietal interactions intensify in the delta band, weaving bridges between distant cortical hubs. Delta and theta bands show increased long-range interactions, particularly in frontoparietal and fronto-parieto-occipital regions.
The flow of information within the brain also changes, as activity shifts from the back of the brain, reflecting taking in and processing outside information, to a concentration of excitatory activity in the front of the brain where the brain reprocesses internal information. During both wakefulness and sleep, information primarily flows from the back of the brain (posterior) to the front (anterior) at higher frequencies. The delta frequency exhibits a hierarchical flow towards the bilateral frontal regions, involving both direct and indirect pathways. This suggests there is a coordinated organization of information flow with sleep onset.
The transition to sleep is also accompanied by a global increase in cortical excitation relative to inhibition, reflecting this shift in the information flow. Using a Neural Mass Model (NMM), the researchers observed spatially heterogeneous patterns, with higher excitation-to-inhibition ratios over bilateral frontal lobes during light NREM compared to wakefulness.
This study marks a significant step forward in our understanding of sleep physiology, unraveling the intricate neural processes guiding the transition from wakefulness to light NREM sleep. These new insights into the complex dynamics of brain networks highlights the coordinated interplay of frequency-specific oscillations, information flow, and excitation-inhibition balance. As we continue to decode the mysteries of the sleeping brain, the knowledge gained from studies like these may pave the way for innovative approaches to enhance sleep quality and address sleep-related disorders.
Study authors include neurologists Joline Fan, MD, MS, Kamalini Ranasinghe, MBBS, PhD, Keith Vossel, and Heidi Kirsch, MD, MS, with psychiatrist Andrew Krystal, MD, and imaging scientists Kiwamu Kudo, Parul Verma, PhD, Hiro Morise, Anne Findlay, Ashish Raj, PhD, and Srikantan Nagarajan, PhD.
