Precision Medicine and Radiology: Using Groundbreaking Research to Detect Cancer Driver MYC
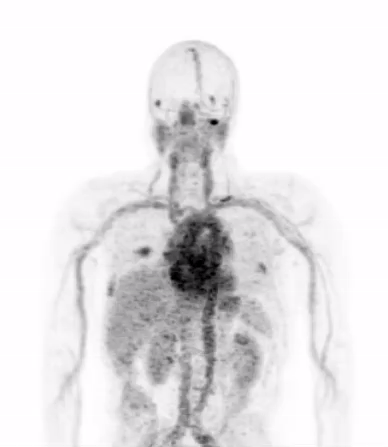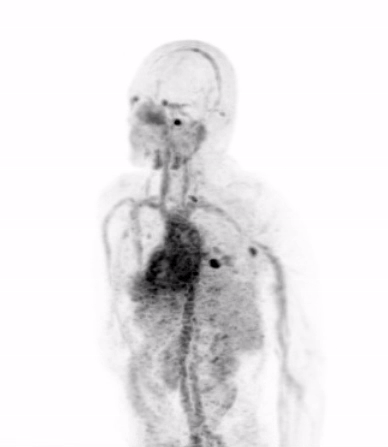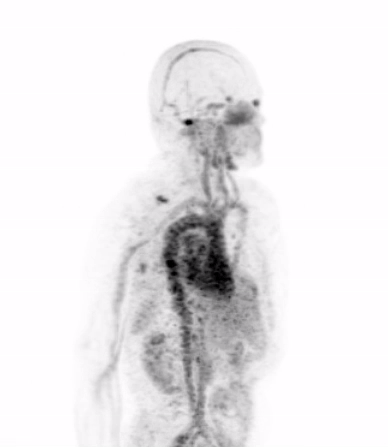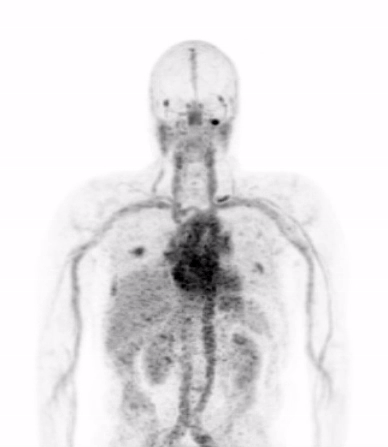 In the current era of precision medicine, the oncology community of medical professionals understands that the biological features of a patient’s tumor must be characterized to ensure that the appropriate treatments are being chosen to inhibit the dominant driver of that patient’s cancer. Today’s gold standard for studying tumor biology in patients is biopsy, or the surgical removal of a piece of tumor for detailed analysis in the laboratory.
In the current era of precision medicine, the oncology community of medical professionals understands that the biological features of a patient’s tumor must be characterized to ensure that the appropriate treatments are being chosen to inhibit the dominant driver of that patient’s cancer. Today’s gold standard for studying tumor biology in patients is biopsy, or the surgical removal of a piece of tumor for detailed analysis in the laboratory.
However, tumor biopsy is generally only applied to early stage cancer that has not metastasized. Applying biopsy to study metastatic cancer is challenging for several reasons. First, biopsy requires invasive procedures to access tumor tissue, which creates severe discomfort for the patient. Second, metastatic cancer lesions are typically very small, and the biopsy procedure is not guaranteed to result in tumor tissue that can be analyzed. Lastly, individual cancer metastases can evolve separately over time, so a biopsy and analysis of one metastasis in a patient may not represent the overall landscape of the biology of other metastases.
If biopsy is not a realistic option for patients with advanced cancer, how can physicians characterize the drivers of a patient’s cancer to select the best drug?
Dr. Michael Evans and his colleagues at UCSF recently published a study that suggests carefully designed PET scans can be used to provide this biological information in place of biopsy.
Dr. Evans and his collaborators, most notably Drs. Rahul Aggarwal and Eric Small of the Genitourinary Oncology Service, and Dr. Spencer Behr of the Department of Radiology and Biomedical Imaging, conducted the first study in patients showing that prostate cancer metastases can be imaged with a new PET scan designed to detect only those metastases that are driven by a notorious cancer driver called “MYC”.
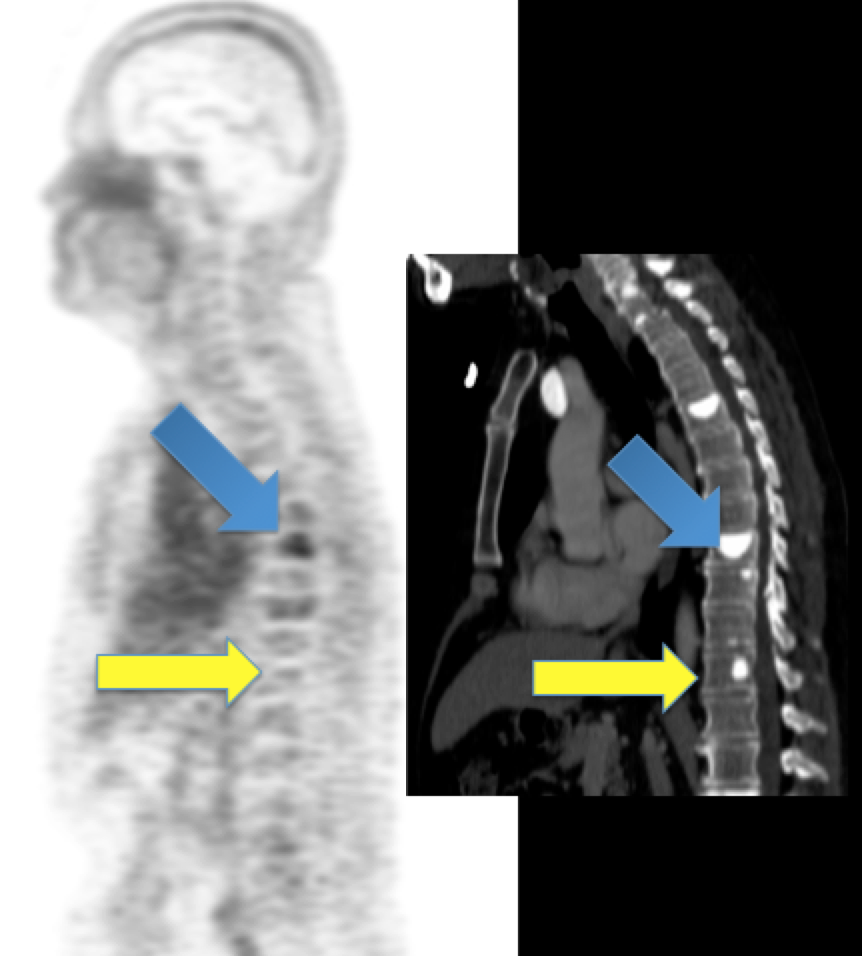
MYC is a protein that is present and conditionally active in almost all cells in the body. However, when MYC is unregulated and hyperactive, it causes many cancers, including common cancers like prostate and breast, as well as rare but very deadly cancers like leukemia and lymphoma. Moreover, even if MYC is not initially hyperactive to cause the onset of cancer, advanced cancer can spontaneously upregulate MYC as a means to escape chemotherapy.
These considerations motivated Dr. Evans and his colleagues to propose that a PET scan that could specifically “light up” cancer cells with hyperactive MYC would be the ideal alternative diagnostic tool to study a patient’s cancer biology in the advanced stages in which biopsy is essentially not performed.
Dr. Evans notes that this technology is timely because “new therapies that suppress MYC are entering clinical trials now at UCSF and other centers for the treatment of prostate cancer and other MYC driven cancers. New experimental therapies are nearly always evaluated in patients that have failed standard treatments and as a result have very advanced cancer. These are precisely the patients to which biopsy cannot be easily applied, and for which a “PET biopsy” is most urgently needed. The new PET scan my colleagues and I developed will allow us to identify which patients have tumors with hyperactive MYC, and this in turn will identify those most likely to respond to drugs that inhibit MYC.”
Is this innovation unique to prostate cancer? Dr. Evans explains that it is not: “MYC is public enemy number one, if you will, in cancer. It contributes to initiation, progression and treatment resistance to some extent for virtually all cancers. We anticipate that having a tool like this new PET scan will dramatically impact treatment selection for many patient populations. Testing this PET scan first in prostate cancer patients is simply a reflection of the exceptional initiative of Drs. Aggarwal and Small to integrate new PET scans into their patients’ care. Since we completed the first human studies in prostate cancer, we have opened trials at UCSF for other MYC driven cancers like liver cancer, lymphoma, and some brain tumors, and we are actively working with other UCSF investigators to open even more.”
Can the approach to developing a PET scan to measure MYC activation be applied to other major biological drivers of cancer? Dr. Evans explains that it can: “Our approach to develop this scan was elegantly straightforward. We used cell biology in preclinical models of cancer to identify a biological event that MYC specifically regulated and could also be imaged with PET. After about two years of validation in preclinical models of cancer, we collected the first patient scan in mid-2014. With the recent advances in the rapid, global profiling of biology regulated by cancer drivers in the laboratory, I can easily visualize how we could have PET scans that specifically measure the activity of the 20 or so major cancer drivers in patients within 10 years. This repertoire of PET scans will be invaluable to understand which driver is responsible for cancer’s escape from drug treatment, as well identifying which therapy is then the most relevant to eliminate the treatment refractory tumor cells.”
Learn more about Dr. Evans’ research and how his lab is using imaging biomarker development to foster precision medicine by clicking here.
Research article, A Feasibility Study Showing [68Ga]Citrate PET Detects Prostate Cancer
