
Shabnam Peyvandi, MD, MAS (right), is a pediatric cardiologist at UCSF Benioff Children’s Hospitals and senior author of the study that found that babies who did not respond to increased oxygen and had a severe type of congenital heart disease had significantly smaller brain volumes. Elizabeth George, MBBS (left), is a neuroradiologist seen consulting with Peyvandi. Photo by Marco Sanchez
Tiny Anatomy, Big Promise: The Rise of Fetal MRI
When UCSF performed its first fetal MRI in 1996, the technology was still largely experimental. Nearly three decades later, fetal MRI has become a vital clinical and research tool. UCSF is now among the most experienced centers in the world in fetal neuroimaging and performs 200-250 scans each year.
That phenomenal growth has been driven by expanding UCSF outreach and education, increasing awareness among OB-GYNs and maternal-fetal medicine specialists, and increasing recognition that MRI can reveal brain details that ultrasound cannot. It has also been fueled by UCSF’s leadership in advancing how fetal brain imaging is performed, interpreted and applied to patient care.
“More and more sites are doing fetal MRI now,” said Orit Glenn, MD, professor and director of Pediatric Neuroradiology at UCSF. “It’s becoming part of the standard clinical evaluation for complicated pregnancies.”
UCSF’s contributions include advances in the interpretation of fetal brain abnormalities, foundational research on normal fetal brain development, and the creation of the Center of Excellence in Fetal Neurology, an interdisciplinary program that brings together neuroradiology, maternal–fetal medicine, fetal treatment specialists, neurology, neurosurgery, and genetics to deliver highly coordinated care.
A Field Still in Its Infancy
Despite this growth, experts emphasize that the field is still young and uniquely challenging.
“These fetal brains are only a few centimeters in size. They are constantly moving, and they are changing rapidly week to week,” said Andreas Rauschecker, MD, PhD, assistant professor of neuroradiology and associate director of the neuroradiology fellowship program.
Duan Xu, PhD, professor of radiology, agrees. “Fetuses are not just smaller versions of adults. Everything about imaging them requires different thinking.”
The ongoing challenges include fetus’s tiny and rapidly evolving anatomy, constant fetal motion and brain structures that change week by week during gestation.
Yet technical advances over the past three decades are transforming what is possible.
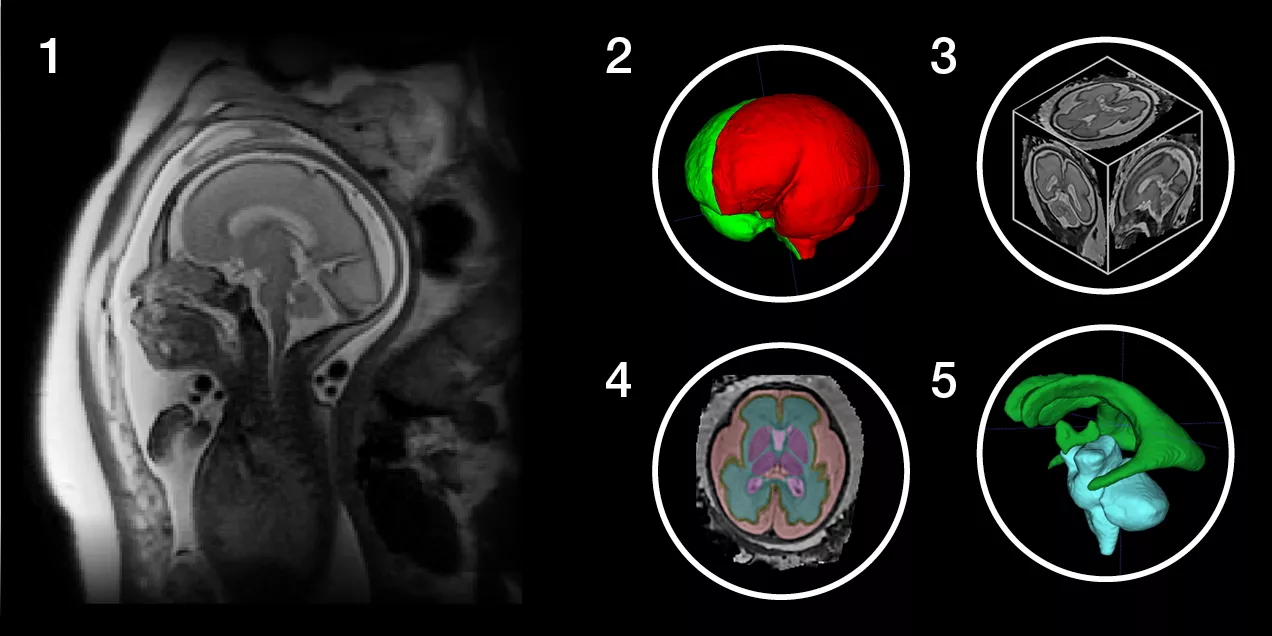
Historically, fetal MRI interpretation has relied heavily on expert qualitative assessment. Now, the field is shifting toward quantitative imaging, which involves developing objective measures that can be used more broadly and consistently.
“Techniques like T2* mapping may not sound flashy,” said Jing Liu, PhD, professor of radiology, “but when they work reliably in fetal brains, the clinical value is enormous.”
Using specialized MRI, T2* mapping shows variations in oxygen levels and tissue properties across organs. In addition, UCSF now routinely performs fetal MRI at 3 Tesla (3T), a higher magnetic field strength that allows clinicians to see finer details of brain development and detect more subtle abnormalities than ever before.
“Our goal is to make advanced imaging fast, robust, and practical enough to actually be used in real clinical care,” she said.
Artificial intelligence is increasingly being used as a research tool to better understand and expand the benefits of fetal MRI, said Rauschecker. “AI gives us the opportunity to extract much more information from scans and potentially spread that expertise beyond centers like UCSF,” said Rauschecker.
A recent study in AJNR co-authored by Rauschecker explored how fetal imaging can inform which patients truly benefit from surgical intervention. The study used AI-based segmentation to analyze ventricular size in normal fetuses and fetuses with ventriculomegaly (enlarged brain ventricles). The researchers built a normative reference database from these scans and compared imaging patterns with real outcomes to determine which babies ultimately required shunting after birth. Fetuses who require postnatal shunting are likely the same ones who would benefit most from in utero intervention.
“By using AI we can begin to predict which babies will truly benefit from surgery and which may not need intervention at all,” Rauschecker said.
Seeing What Was Once Invisible
MRI reveals significantly more detail about fetal brain structure than ultrasound alone. This allows clinicians to detect abnormalities that are extraordinarily subtle but can be clinically meaningful.
One example is periventricular nodular heterotopia (PVNH), a condition involving tiny disruptions in how neurons migrate during brain development. These abnormalities may appear as only a few pixels on a fetal MRI scan and must be confirmed across multiple imaging planes. The condition can be associated with seizure risk later in life.
“Some of these findings are only a few pixels on a single slice,” said Yi Li, MD, associate professor and neuroradiology fellowship director. “We have to confirm them in multiple planes to be sure. It’s remarkable that we can see them at all.”
Another example involves COL4A gene mutations, which affect the basement membrane and can cause fetal brain hemorrhage or infarction. UCSF researchers have helped characterize the spectrum of imaging findings for these disorders in utero, improving diagnostic accuracy and counseling.
Fetal imaging is also helping clinicians identify risk earlier and more precisely in complex conditions. In a recent JAHA study co-authored by Jing Liu, PhD, and Duan Xu, PhD, clinicians used advanced fetal brain MRI alongside a simple maternal oxygen test in babies with congenital heart disease to determine which fetuses were at highest risk for brain injury and later developmental differences. Babies who did not show normal blood-flow responses in utero were more likely to have smaller brain volumes and early brain injury after birth. The findings illustrate how fetal imaging is beginning to shift care earlier and open the door to future prenatal interventions.
But fetal imaging is not only about identifying problems. It can also provide reassurance. “Sometimes the most powerful thing we can tell a family is that everything looks normal,” said Elizabeth George, MBBS, neuroradiologist. Applying advanced imaging to fetal populations can unlock information that’s simply not available any other way.
Understanding what fetal imaging findings truly mean requires long-term follow-up from before birth through childhood. “That’s why longitudinal data is so critical,” Glenn said. “It takes years to build that knowledge.”
“If we can detect something early and influence a child’s developmental trajectory, the impact can last 60, 70, even 80 years,” Xu said.
Prenatal Genetic Testing and Looking Ahead
As prenatal genetic testing becomes more advanced and more accessible, researchers are increasingly able to pair genetic findings with fetal MRI patterns. This combination can improve diagnostic accuracy and help identify certain conditions earlier in pregnancy.
“The interplay between genetics and brain development is where the field is really going to grow next,” said Glenn.
Earlier diagnosis is also expanding what may be possible for treatment. UCSF is already a leader in developing in utero therapies for select genetic and congenital conditions, and imaging plays a central role in that work by helping clinicians identify candidates and monitor response.
“We’re starting to see movement toward treating certain genetic conditions before birth,” Glenn said. “Fetal MRI will be key to both detecting abnormalities and tracking whether therapies are working.”
Advancing this kind of work depends on close collaboration across specialties. At UCSF, that interdisciplinary approach led Glenn to partner with fetal treatment center leadership and child neurology to establish the Center of Excellence in Fetal Neurology, which recognizes the advanced level of care made possible through tightly integrated expertise.
“Fetal care demands true teamwork,” she said. “Our ability to integrate subspecialists from radiology, neurology, maternal–fetal medicine, surgery, neonatology, genetics, and fetal therapy allows us to provide comprehensive, highly specialized care.”
That collaborative model is also shaping the field beyond UCSF. Many former neuroradiology fellows have gone on to establish or expand fetal MRI programs nationwide, helping spread expertise and influence the future of the field.
References
Zhou M, Rajan SA, Nedelec P, Bayona JB, Glenn O, Gupta N, Gano D, George E, Rauschecker AM. Prediction of cerebrospinal fluid intervention in fetal ventriculomegaly via AI-powered normative modelling. AJNR Am J Neuroradiol. 2025 Sep 16. doi: 10.3174/ajnr.A9000. Epub ahead of print. PMID: 40957691.
Taleb M, Liu J, Xu D, Zhao Y, Moon-Grady AJ, McQuillen PS, Peyvandi S. Fetal Cerebrovascular Response to Maternal Hyperoxia Testing and Association with Brain Growth and Postnatal Brain Injury in Congenital Heart Disease. JAHA Journal of the American Heart Association. 2025 Aug 5. 14(15). doi.org/10.1161/JAHA.125.042014