Precision Oncology: Fast-Tracking Cancer Treatment with Co-clinical Trials and Metabolic MRI
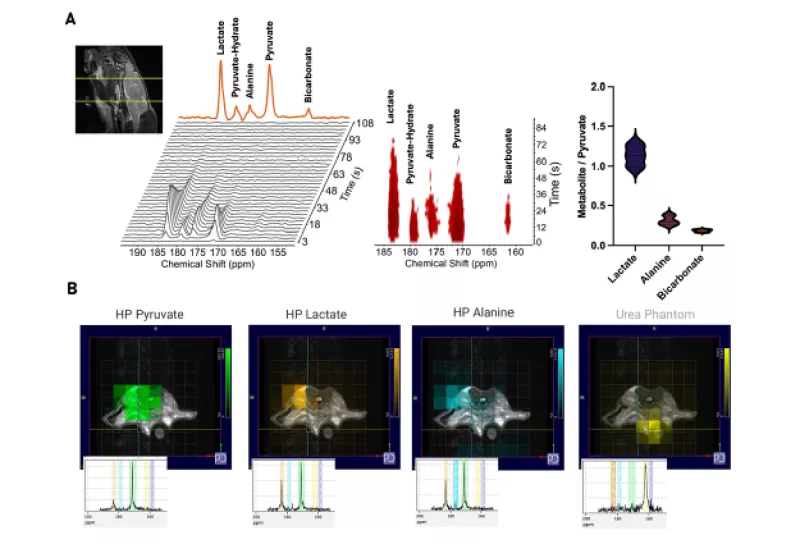
While clinicians use standard MRI and CT imaging to measure the size and distribution of tumors, metabolic imaging offers new ways to understand how cancer therapies work on a cellular level. Renuka Sriram, PhD, associate professor and director of the pre-clinical core in the Department of Radiology and Biomedical imaging at UCSF, uses hyperpolarized metabolic MRI to analyze how cancers and other diseases progress and respond to treatment, using co-clinical trials and patient derived models for the fastest and most accurate answers.
To do this, Sriram tracks hyperpolarized agents as cells metabolize it into downstream metabolic products. Imaging the flow of the molecules shows where metabolically active cancer tumors are growing or regressing. Sriram and her team focus on aggressive cancers with high metabolic activity, particularly the neuroendocrine small cell phenotype of prostate cancer, which is often found in the bone and liver with poor prognosis and are hard to treat.
New Technology Improves Metabolic MRI
Until recently, metabolic imaging was rarely used in co-clinical trials. Polarizers, the instruments used to boost the signal of the 13C enriched agents, have a pre-preparation cycle of 90 minutes. Any technical failure, such as a catheter slipping out of an anesthetized animal, meant the team had to wait another 90 minutes to administer a new dose.
That challenge has recently been addressed, now that UCSF is home to the first of a new generation of polarizers which can prepare an imaging agent in just 30 minutes. Sriram and her colleagues are the first to ever use this technology in vivo. Sriram expects that this ease, speed and physiologically relevant concentrations will significantly expand metabolic MRI in pre-clinical research and ultimately improve logistics for clinical application. Sriram observes, "We’ve merely scratched the surface of hyperpolarized carbon imaging, and the potential is essentially limitless in terms of accelerating our work with co-clinical trials using metabolic MRI technology.”
Watching Metabolism Everywhere
The potential of co-clinical trials using hyperpolarized metabolic MRI extends far beyond cancer. The technology can be used to distinguish inflammation such as in arthritis or infection by detecting bacteria specific metabolic pathways. The bioenergetics of improved understanding of neurotransmission are also relevant for Alzheimer’s disease, stroke, and multiple sclerosis.
Sriram’s work on metabolic MRI and co-clinical trials can help quickly confirm drug delivery, monitor its uptake at the target, watch the pharmacodynamic effects, and characterize a tumor's function and heterogeneity with unprecedented detail. By applying metabolic MRI to co-clinical trials, Sriram and her lab are compressing the timeline for bringing effective treatments to patients.
What are Co-Clinical Trials?
Concurrently or prospectively conducted clinical and pre-clinical trials in which—
- Animals are selected to match the patient cohorts
- Treatment and monitoring protocols are the same
- Allows real time data sharing to optimize patient strategies
- Enables rapid mechanism discovery